Rb (Rubidium) is an element with position number 37 in the periodic table.
Located in the V period. Melting point: 39 ℃. Density: 1.53 g/cm3.
Electronic configuration of the Rubidium atom in ascending order of orbital energies:
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1
Electronic configuration of the Rubidium atom in ascending order of the levels:
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 5s1
Reduced electronic configuration Rb:
[Kr] 5s1
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1
Electronic configuration of the Rubidium atom in ascending order of the levels:
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 5s1
Reduced electronic configuration Rb:
[Kr] 5s1
Below is the electronic diagram of the Rubidium atom
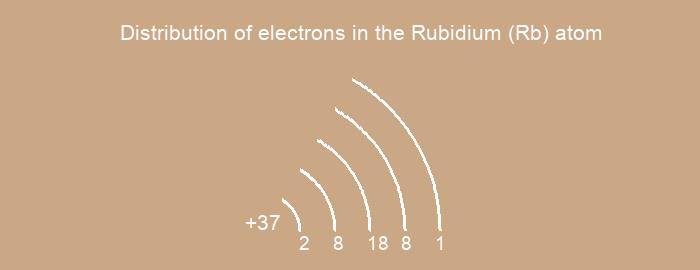
 Distribution of electrons over energy levels in the Rb atom
Distribution of electrons over energy levels in the Rb atom
1-st level (K): 2
2-st level (L): 8
3-st level (M): 18
4-st level (N): 8
5-st level (O): 1

1-st level (K): 2
2-st level (L): 8
3-st level (M): 18
4-st level (N): 8
5-st level (O): 1

Valence electrons of Rubidium
The number of valence electrons in a Rubidium atom - 1.
Below are their quantum numbers (N - energy, L - angular momentum, M - magnetic moment, S - spin )
Below are their quantum numbers (N - energy, L - angular momentum, M - magnetic moment, S - spin )
| Orbital | N | L | M | S |
|---|---|---|---|---|
| s | 5 | 0 | 0 | +1/2 |
Oxidation states of Rubidium: +1
Other elements
Electronic configurations of other elements
Krypton (Kr) electron configurationStrontium (Sr) electron configuration
Electronic configuration table
