Pb (Lead) is an element with position number 82 in the periodic table.
Located in the VI period. Melting point: 327.5 ℃. Density: 11.34 g/cm3.
Electronic configuration of the Lead atom in ascending order of orbital energies:
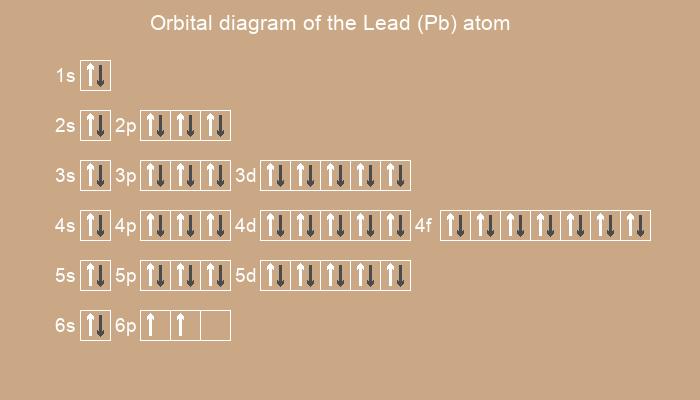
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p2
Electronic configuration of the Lead atom in ascending order of the levels:
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d10 6s2 6p2
Reduced electronic configuration Pb:
[Xe] 4f14 5d10 6s2 6p2
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p2
Electronic configuration of the Lead atom in ascending order of the levels:
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d10 6s2 6p2
Reduced electronic configuration Pb:
[Xe] 4f14 5d10 6s2 6p2
Below is the electronic diagram of the Lead atom
 Distribution of electrons over energy levels in the Pb atom
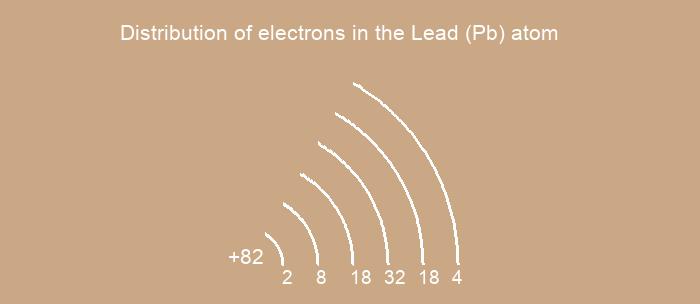
Distribution of electrons over energy levels in the Pb atom
1-st level (K): 2
2-st level (L): 8
3-st level (M): 18
4-st level (N): 32
5-st level (O): 18
6-st level (P): 4

1-st level (K): 2
2-st level (L): 8
3-st level (M): 18
4-st level (N): 32
5-st level (O): 18
6-st level (P): 4

Valence electrons of Lead
The number of valence electrons in a Lead atom - 4.
Below are their quantum numbers (N - energy, L - angular momentum, M - magnetic moment, S - spin )
Below are their quantum numbers (N - energy, L - angular momentum, M - magnetic moment, S - spin )
| Orbital | N | L | M | S |
|---|---|---|---|---|
| s | 6 | 0 | 0 | +1/2 |
| s | 6 | 0 | 0 | -1/2 |
| p | 6 | 1 | -1 | +1/2 |
| p | 6 | 1 | 0 | +1/2 |
Oxidation states of Lead: -4, +2, +4
Other elements
Electronic configurations of other elements
Thallium (Tl) electron configurationBismuth (Bi) electron configuration
Electronic configuration table
